SURFACE TREATMENT
Surface treatment refers to a group of processes used to alter the surface of a material, typically metal, to improve its appearance, performance, or durability. These treatments are crucial in manufacturing and engineering to enhance corrosion resistance, wear resistance, adhesion, electrical conductivity, or aesthetic quality.
Purpose of Surface Treatment:
- Protect against corrosion (e.g., rust on steel)
- Improve adhesion before painting or coating
- Enhance appearance (shine, color, texture)
- Increase wear resistance and hardness
- Modify electrical or thermal conductivity
- Make surfaces easier to clean or more hygienic
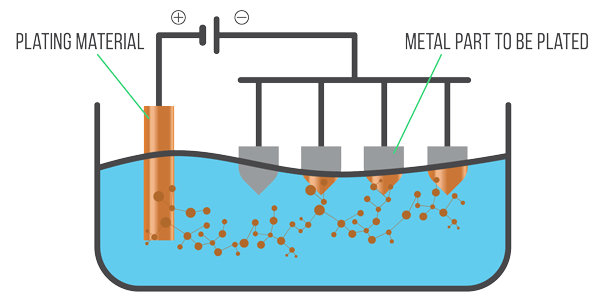
The Electroplating Process:
The process typically involves the following components:
- Anode: This is the positively charged electrode. It's usually made of the metal that will be deposited onto the workpiece. During electroplating, the anode metal dissolves into the electrolyte solution as positively charged ions (cations). In some cases, an inert anode (like carbon or platinum) is used, and the metal ions in the solution are replenished from other sources.
- Cathode: This is the negatively charged electrode, which is the workpiece or object to be plated. The positively charged metal ions in the electrolyte are attracted to the cathode.
- Electrolyte: This is a liquid solution containing dissolved salts of the metal to be deposited. It facilitates the flow of electric current and provides the metal ions needed for plating. Other chemicals may be added to the electrolyte to improve conductivity, control pH, enhance brightness, and ensure a uniform deposit.
Power Source: A direct current (DC) power source is connected to the anode and cathode, providing the electrical energy needed for the process.
Electroless plating, also known as autocatalytic plating or chemical plating, is a method of depositing a thin layer of metal onto a substrate without the use of an external electric current. Unlike electroplating, which relies on an electric field to drive the deposition, electroless plating uses a chemical reduction reaction in solution.
The Electroless Plating Process:
- Surface Preparation: Thorough cleaning of the substrate is crucial to remove any dirt, oil, oxides, or other contaminants that could hinder adhesion of the plated layer. This often involves a series of chemical baths, including degreasing, etching, and rinsing steps. Internal stresses in the substrate can also affect the plating.
- Activation: The cleaned surface is often treated with an acidic solution to activate it, removing any oxide film and revealing a fresh metal surface. For non-conductive materials, a pre-treatment with a catalyst, such as palladium, is necessary to initiate the deposition.
- Electroless Plating Bath: The substrate is immersed in a chemical solution containing:
- Metal ions: A source of the metal to be deposited (e.g., nickel salts like nickel sulfate).
- Reducing agent: A chemical that donates electrons to the metal ions, causing them to be reduced to their metallic form and deposit on the substrate (e.g., sodium hypophosphite for nickel plating, formaldehyde for copper plating).
- Complexing agents: These help to increase the solubility of the metal salts and prevent precipitation by controlling the concentration of free metal ions. They also act as buffers to maintain the bath's pH.
- Stabilizers: These prevent the spontaneous decomposition of the plating bath and control the rate of deposition.
- Buffers: Maintain the desired pH of the bath, as the chemical reaction often produces byproducts that can change the pH.
- Accelerators (optional): Can be added to counteract the reduction in plating rate caused by complexing agents.
- Brighteners (optional): Certain organic or inorganic compounds can be added to improve the surface finish.
- Surfactants (optional): Help to ensure a hydrophilic surface, reducing pitting and staining.
- Autocatalytic Reaction: The deposition of metal is autocatalytic, meaning that once a thin layer of the metal is deposited, the metal itself catalyzes further deposition. The reducing agent reacts with the metal ions at the substrate's surface, causing the metal to plate out.
- Post-Treatment: After plating, the component is typically rinsed, dried, and may undergo further post-treatment like heat treatment to enhance adhesion, corrosion resistance, and other properties. This can include baking or passivation.
Chemical treatment in the context of surface finishing refers to a range of processes that utilize chemical reactions to alter the surface properties of a material, typically metals. These treatments aim to enhance characteristics like corrosion resistance, wear resistance, paint adhesion, electrical conductivity, or aesthetic appearance. Unlike coating processes that add a layer of a different material, chemical treatments involve a reaction with the existing surface of the material, forming a new layer or modifying its composition.
Here's a breakdown of common types of chemical treatments:
- Chemical Conversion Coatings:
These treatments involve a chemical reaction between the metal surface and a specific solution, resulting in the formation of a thin, non-metallic, and often protective layer that is chemically bonded to the metal.
- Phosphating: Primarily used on steel and iron, this process creates a layer of insoluble metal phosphates (iron, zinc, or manganese phosphate). The porous nature of the phosphate coating provides excellent adhesion for paints, powder coatings, and lubricants, and offers mild to moderate corrosion resistance. Different types of phosphating are chosen based on the desired level of corrosion protection and subsequent finishing processes.
- Chromate Conversion Coating (Chem Film/Alodine): Commonly applied to aluminum alloys, as well as zinc and steel, this process forms a thin, gel-like layer containing chromium compounds. It offers excellent corrosion resistance and good adhesion for paints and adhesives. The color can range from clear to iridescent yellow or gold, depending on the specific chemistry. Trivalent chromium coatings are increasingly used due to environmental concerns associated with hexavalent chromium.
- Black Oxide (Bluing): Used mainly on ferrous metals (steel and iron), this process forms a thin layer of black iron oxide (magnetite) on the surface. It provides a mild level of corrosion resistance and is often used for its aesthetic black finish and to improve paint adhesion. A post-treatment with oil or wax is typically required to enhance corrosion protection.
- Pickling:
This is an acid treatment used to remove surface impurities, such as scale (oxides formed during high-temperature processing like welding or heat treatment), rust, and other contaminants from metals like steel and stainless steel. Pickling etches the metal surface, leaving it clean and ready for further processing. Common pickling acids include hydrochloric acid, sulfuric acid, and nitric acid (often in mixtures, especially for stainless steel).
- Passivation:
Primarily used for stainless steel and other corrosion-resistant metals like titanium, passivation involves treating the surface with a mild oxidizing agent, such as nitric acid or citric acid solutions. This process removes free iron and other surface contaminants, allowing the natural chromium oxide layer (which provides stainless steel's corrosion resistance) to form and become more stable and effective. Passivation does not significantly change the appearance or dimensions of the part.
- Etching:
Chemical etching uses acids or alkaline solutions to selectively remove material from a metal surface. This can be done for various purposes, including:
- Cleaning: Removing surface contaminants.
- Creating textures or patterns: For decorative or functional purposes (e.g., increasing adhesion).
- Preparing for other surface treatments: Creating a micro-roughened surface for better coating adhesion.
- Dimensional control: In specific applications.
- Electroless Plating:
While often categorized separately, electroless plating is a chemical reduction process that deposits a thin layer of metal onto a substrate without using an external electric current. The deposition is achieved through a controlled chemical reaction in the plating solution. Common electroless plating metals include nickel, copper, and gold. It offers uniform coating thickness even on complex shapes and non-conductive materials (after activation).
- Other Chemical Treatments:
- Chelating Agents: Chemicals used to remove specific surface contaminants by forming stable, water-soluble complexes with metal ions.
- Alkaline Cleaning: Used to remove organic contaminants like oils and grease from metal surfaces.
- Acid Cleaning: Used to remove inorganic contaminants like rust and scale.

Anodic Oxidation is an electrochemical process that forms an oxide layer on the surface of a metal, typically aluminum but also applicable to other metals like titanium, zinc, and magnesium. The metal part is made the anode (positive electrode) in an electrolytic cell, and when a current is passed through the electrolyte, an oxide layer grows on the metal's surface. This layer is integral to the metal, not just a coating, which gives it excellent adhesion and durability.
Here's a deeper dive into anodic oxidation:
The Process:
- Pre-treatment: The metal part is thoroughly cleaned to remove dirt, grease, and any existing oxides. This may involve degreasing, etching, and desmutting.
- Anodizing Bath: The cleaned part is immersed in an electrolytic solution, most commonly sulfuric acid, chromic acid, or oxalic acid. The specific acid and its concentration influence the properties of the resulting oxide layer. The bath temperature is also carefully controlled.
- Electrical Setup: The aluminum part acts as the anode, and a cathode (usually made of lead or aluminum) is also immersed in the electrolyte. A direct current (DC) power supply is connected to the electrodes.
- Oxidation: When the electric current flows through the electrolyte, oxygen ions are released at the surface of the anode (the aluminum part). These oxygen ions react with the aluminum atoms to form aluminum oxide (Al₂O₃). The oxide layer grows from the surface inwards.
- Rinsing: After the desired thickness of the oxide layer is achieved, the part is removed from the anodizing bath and thoroughly rinsed to remove any residual electrolyte.
- Sealing (Optional but Recommended): The anodic oxide layer is porous. Sealing involves immersing the anodized part in a hot water bath (often with additives like nickel acetate) to hydrate the aluminum oxide. This closes the pores, improving corrosion resistance and making the surface less susceptible to staining.
- Coloring (Optional): Before sealing, the porous anodic layer can be dyed with various organic or inorganic dyes to achieve a wide range of colors for aesthetic or identification purposes.


Hot dipping is a surface treatment process where a base material, most commonly steel or iron, is immersed in a bath of molten metal to create a protective coating. The most prevalent type of hot dipping is hot-dip galvanizing, where the molten metal is zinc. However, the principle can be applied using other metals like aluminum (hot-dip aluminizing) or tin (hot-tinning).
Since "hot dipping" is often synonymous with "hot-dip galvanizing" due to its widespread use, the following explanation will primarily focus on this process while also touching upon other hot-dipping applications.
Hot-Dip Galvanizing:
Process:
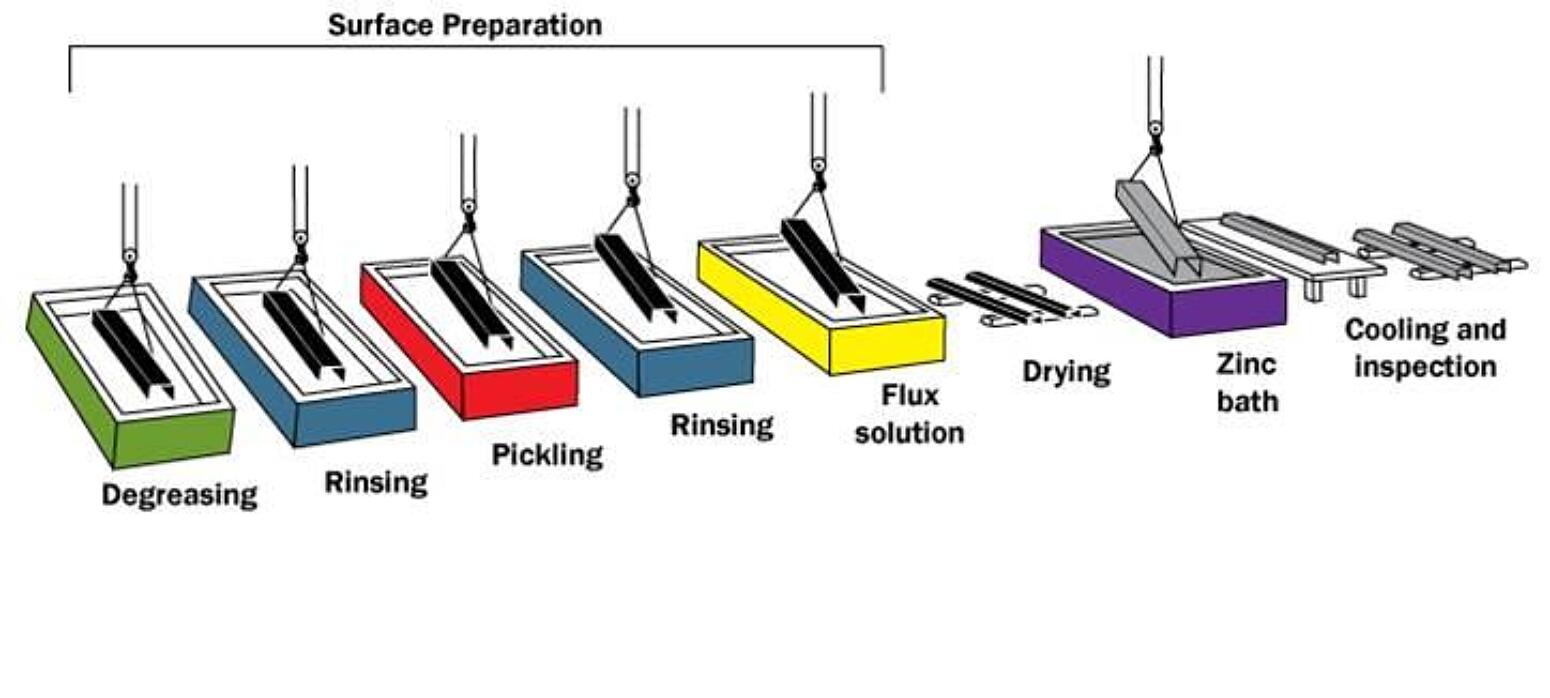
Hot-dip galvanizing involves several key steps:
- Surface Preparation: This is crucial for a quality coating. The steel or iron goes through a rigorous cleaning process to remove:
- Degreasing: Removal of oil, grease, and organic contaminants using alkaline solutions, mild acids, or biological cleaning baths.
- Rinsing: To wash off the cleaning solutions.
- Pickling: Immersion in an acidic solution (usually hydrochloric or sulfuric acid) to remove mill scale, rust, and iron oxides. Abrasive blasting can also be used.
- Rinsing: Again, to remove the pickling solution.
- Fluxing: Application of a flux solution (often zinc ammonium chloride) to the cleaned steel. This removes any remaining oxides and creates a protective layer to promote the reaction between the steel and molten zinc. The flux is usually dried onto the steel.
- Galvanizing: The prepared steel is then slowly immersed into a bath of molten zinc (at a temperature of around 450°C or 842°F) for a specific amount of time until the temperature of the steel equals that of the bath. During this immersion, a metallurgical reaction occurs between the iron in the steel and the zinc, forming a series of zinc-iron alloy layers and an outer layer of pure zinc.
- Withdrawal: The steel is carefully removed from the molten zinc bath. Excess zinc is removed by draining, vibrating, or using air knives.
- Cooling: The galvanized steel is then cooled, either by quenching in a passivation solution (to prevent the formation of white rust) or by air cooling.
- Inspection: The final coating is inspected for thickness, uniformity, and any defects. Magnetic thickness gauges are commonly used to verify the coating thickness meets specifications.
Vacuum plating, also known as physical vapor deposition (PVD), is a surface treatment process carried out in a vacuum environment to deposit thin films of various materials onto a substrate. The goal is to enhance the substrate's surface properties, such as its appearance, durability, corrosion resistance, hardness, or electrical conductivity.
In vacuum plating, the material to be deposited is vaporized (either physically or chemically) within a vacuum chamber and then condenses onto the surface of the workpiece. The vacuum environment is crucial as it reduces the presence of air and other gases, allowing for a purer and more controlled deposition process.
Main Types of Vacuum Plating:
Vacuum plating encompasses several specific techniques, each with its own mechanism for vaporizing the coating material:
- Vacuum Evaporation (Thermal Evaporation):
- Process: The coating material (in solid or liquid form) is heated in a high vacuum until it evaporates. The vaporized atoms then travel through the vacuum and condense as a thin film on the cooler substrate.
- Methods: Resistance heating, electron beam evaporation.
- Applications: Optical coatings (lenses, mirrors), reflective coatings, decorative coatings on plastics and metals.
- Sputtering:
- Process: A target material (the material to be deposited) is bombarded with high-energy ions (usually inert gases like argon) in a vacuum chamber. This bombardment causes atoms or clusters of atoms to be ejected from the target. These ejected atoms then travel and deposit as a thin film on the substrate.
- Methods: DC sputtering (for conductive materials), RF sputtering (for non-conductive materials), magnetron sputtering (uses magnetic fields to enhance plasma density and deposition rate).
- Applications: Semiconductor thin films, wear-resistant coatings on tools, decorative coatings, thin films for solar cells and displays.
- Ion Plating:
- Process: This technique combines aspects of both evaporation and sputtering. The coating material is vaporized (usually by evaporation or sputtering), and the vaporized atoms are then ionized by a plasma within the vacuum chamber. These ions are accelerated towards the substrate by an electric field, resulting in a denser and more adherent coating due to the high kinetic energy of the impinging ions.
- Methods: Reactive ion plating (uses reactive gases to form compound coatings like nitrides or oxides), bias sputtering (applies a negative bias to the substrate to attract ions).
- Applications: Hard and wear-resistant coatings on cutting tools, decorative coatings with high durability, corrosion-resistant coatings.
Other Vacuum Deposition Techniques (often included under the broader term "vacuum plating"):
- Chemical Vapor Deposition (CVD): While sometimes distinguished from PVD, CVD is a vacuum-based process where gaseous precursors react chemically at the substrate surface to form a solid thin film.
- Atomic Layer Deposition (ALD): A highly controlled CVD technique that deposits materials one atomic layer at a time, offering exceptional thickness control and conformality.


Painting is a widely used surface treatment process that involves applying a liquid or powdered coating of pigmented material to a substrate (such as metal, wood, plastic, or masonry) for various purposes, including:
- Aesthetics: Providing color, gloss, texture, and decorative effects.
- Protection: Shielding the substrate from corrosion, weathering, chemicals, abrasion, and other forms of damage.
- Functionality: Imparting specific properties like electrical insulation, conductivity, reflectivity, or slip resistance.
Painting is a versatile process with a wide range of techniques and paint formulations tailored to different applications and performance requirements.
The Painting Process (General Steps):
While specific steps can vary depending on the paint type, application method, and substrate, the general process often includes:
- Surface Preparation: This is a crucial step for ensuring good adhesion and the longevity of the paint job. It involves:
- Cleaning: Removing dirt, dust, grease, oil, rust, mill scale, and other contaminants. Methods include washing, degreasing with solvents or alkaline solutions, abrasive blasting (sandblasting, shot blasting), and chemical cleaning (acid or alkaline etching).
- Repairing Imperfections: Filling holes, cracks, and dents with fillers or body putty, and sanding them smooth.
- Priming (Optional but Recommended): Applying a primer coat to improve adhesion, provide corrosion resistance, seal porous surfaces, and create a uniform base for the topcoat. Different primers are formulated for specific substrates and topcoat types.
- Sanding: Creating a slightly rough surface (keying) for better mechanical adhesion of subsequent coats.
- Paint Application: The chosen paint is applied to the prepared surface using various methods:
- Brushing: Suitable for small areas, intricate details, and touch-ups. Provides good control but can leave brush strokes.
- Rolling: Efficient for covering large, flat surfaces. Different roller nap lengths are used for various paint types and surface textures.
- Spraying: Provides a smooth, even finish over large or complex surfaces. Different types of spray guns are used, including air spray, airless spray, and HVLP (High Volume Low Pressure) spray guns.
- Dipping: Parts are immersed in a tank of paint, ensuring complete coverage, especially for complex shapes. Often used for primers or single-coat applications.
- Electrostatic Painting: A charged paint is sprayed onto an oppositely charged workpiece, improving transfer efficiency and coverage, especially for complex shapes. Commonly used in industrial applications.
- Powder Coating: A dry, free-flowing powder is electrostatically applied and then cured in an oven, creating a durable and often decorative finish.
- Drying and Curing: After application, the paint needs to dry and cure to form a hard, durable film.
- Air Drying: The paint dries naturally through evaporation of solvents and/or chemical reactions with the air.
- Force Drying: Heat lamps or ovens are used to accelerate the drying process.
- Curing: Some paints, especially those with thermosetting resins (like epoxies or urethanes), require a specific temperature and time to undergo a chemical cross-linking reaction that hardens the coating.
- Finishing (Optional): Additional steps may be taken to achieve the desired final appearance or performance:
- Sanding or Rubbing: To smooth out imperfections or adjust the gloss level.
- Polishing: To achieve a high-gloss finish.
- Clear Coating (Varnishing or Lacquering): Applying a transparent topcoat for added protection and gloss.
Types of Paints and Coatings:
A vast array of paint formulations exists, categorized by their binder (the film-forming component), solvent (the liquid carrier), and pigments (the coloring agents):
- Alkyd Paints: Oil-based paints known for good adhesion, durability, and relatively low cost. Used for general-purpose applications on wood and metal.
- Latex (Acrylic) Paints: Water-based paints offering good flexibility, color retention, and ease of cleanup. Popular for interior and exterior walls.
- Epoxy Paints: Two-part systems that provide excellent hardness, chemical resistance, and adhesion. Used in industrial and high-performance applications.
- Polyurethane Paints: Known for their durability, abrasion resistance, and flexibility. Used in automotive finishes, wood coatings, and industrial applications.
- Enamel Paints: Hard, glossy paints that can be oil-based or water-based. Offer good durability and are used for trim, appliances, and metal surfaces.
- Powder Coatings: Solvent-free coatings applied as dry powder and cured with heat, resulting in a tough, durable, and often decorative finish.
- Primers: Formulated to adhere well to the substrate and provide a good base for topcoats. Different primers are designed for specific materials (e.g., metal primers with rust inhibitors).
- Specialty Coatings: Paints formulated for specific purposes, such as heat-resistant coatings, anti-fouling paints, intumescent (fire-resistant) coatings, and textured paints.



Thermal spraying is a versatile surface engineering technique used to apply metallic, ceramic, cermet, or polymeric coatings to a wide range of materials and components. In this process, a coating material (in powder, wire, rod, or liquid form) is heated to a molten or semi-molten state and propelled at high velocity onto a prepared substrate surface. Upon impact, the particles flatten, interlock, and rapidly solidify, forming a mechanically or metallurgically bonded coating.
The broad term "thermal spraying" encompasses various specific processes, each with its unique way of generating heat and propelling the coating material. Here's a breakdown of some common types:
Common Thermal Spraying Processes:
- Flame Spraying (Combustion Spraying): Uses the combustion of a fuel gas (like acetylene, propane, or hydrogen) with oxygen or air to melt the coating material (powder or wire). Compressed air propels the molten particles onto the substrate. It's a relatively low-cost and simple method suitable for less demanding applications.
- Arc Spraying (Wire Arc Spraying): An electric arc is generated between two consumable wires of the coating material, causing them to melt. Compressed air or gas atomizes and propels the molten metal onto the substrate. It offers high deposition rates and is cost-effective for large areas, often used for corrosion protection.
- Plasma Spraying: A high-temperature plasma jet, created by passing an inert gas (like argon or helium) through an electric arc, melts and accelerates the powder coating material. This versatile process can spray a wide range of materials, including high-melting-point ceramics and metals, producing high-quality coatings. Variations include Atmospheric Plasma Spraying (APS), Vacuum Plasma Spraying (VPS), and Low-Pressure Plasma Spraying (LPPS) for specialized applications.
- High-Velocity Oxy-Fuel (HVOF) Spraying: A combustion of fuel (gas or liquid like kerosene) and oxygen in a specially designed chamber creates high-velocity, high-temperature gases. Powdered coating material is injected into this gas stream and propelled at supersonic speeds onto the substrate. HVOF coatings are dense, hard, and have excellent bond strength, making them ideal for wear and corrosion resistance in demanding environments.
- High-Velocity Air-Fuel (HVAF) Spraying: Similar to HVOF but uses compressed air instead of pure oxygen, resulting in lower temperatures and potentially less oxidation of the coating material. HVAF can produce uniform, ductile coatings with good bond strength and is suitable for materials that oxidize easily at higher temperatures.
- Cold Spraying: A unique process where powder particles are accelerated to supersonic speeds using a high-pressure carrier gas (like nitrogen or helium) and propelled onto the substrate without melting. The particles undergo severe plastic deformation upon impact, mechanically bonding to the surface. Cold spraying is advantageous for materials sensitive to high temperatures and can produce dense, low-oxide coatings.
- Detonation Spraying (D-Gun): A mixture of fuel (acetylene) and oxygen is detonated in a water-cooled barrel, heating and propelling powder coating material at extremely high velocities. This pulsed process creates very high-quality, dense, and well-bonded coatings.
- Spray and Fuse: A two-step process where a powder coating is first thermally sprayed onto the substrate, followed by heating with a flame or furnace to melt and fuse the coating to the base material, creating a metallurgical bond for enhanced wear and abrasion resistance.
- Warm Spraying: A modification of HVOF where nitrogen is added to lower the combustion gas temperature. It operates at temperatures between HVOF and cold spray, offering a balance of properties and potentially higher coating efficiency than cold spray for certain materials.
Applications of Thermal Spraying:
Thermal spraying is employed across a vast spectrum of industries to enhance component performance and longevity:
- Aerospace: Coating turbine blades, landing gear components, and other critical parts for wear, corrosion, and thermal barrier protection.
- Automotive: Applying wear-resistant coatings to engine components, exhaust systems, and decorative finishes.
- Oil and Gas: Protecting pipelines, offshore platforms, and downhole tools from corrosion and wear.
- Power Generation: Coating boiler tubes, turbine components, and heat exchangers for corrosion and high-temperature resistance.
- Manufacturing: Repairing and refurbishing worn machinery, applying wear-resistant coatings to tools and dies, and enhancing the surface properties of various components.
- Medical: Applying biocompatible coatings to implants and surgical instruments.
- Electronics: Creating conductive or insulating layers and EMI/RFI shielding.
- Infrastructure: Protecting bridges, buildings, and other structures from corrosion.
- Marine: Coating ship hulls and offshore structures for corrosion and fouling resistance.
- Textile and Paper Industries: Applying wear and non-stick coatings to rollers and other processing equipment.